Similacrecall Com Why is it remembered Today?
Do you want to reveal the project initiated by an United States research lab? You must study this piece of writing to fully understand the issue in depth.
The most delicate of human beings is the toddlers who we can support by feeding them the essential proteins. Other than natural lactation, a majority of people are typically looking for the use of infant formula to help keep their kids well.
However, on the other hand, yesterday the world’s most renowned laboratory announced the recall of some formulation products due to certain reasons related to Similacrecall.com. Therefore, let’s look deeper into the related details in this piece for the benefit of all internet users.
Introducing Abbott Laboratories
Abbott has an american-based testing lab that provides nutritional products as well as medicines as well as medical devices. The laboratory was established at the time of Wallace Calvin Abbott. It is now thriving in more than 160 countries.
What is it and how does it work?
We found out that its primary job is to research infant formulas prior to launch. Recently, we discovered that Abbott labs have been recalled for some nutritional products. Let’s move onto the site for complete information.
Analyzing Similacrecall.com
On February 17, 2022 On the 17th February of 2022, the Abbott testing center announced that it would recall infant powders from Similac, EleCare, and Alimentum. Through the process they discovered that there was Cronobacter Sakazakii, however, the threads from Salmonella Newport were missing.
As per the website it is clear that none of the products were affected by the pathogens, so they are continuing to study. Additionally, they are mainly tracing those products that have expiration dates of 1 April 2022.
If you are interested in obtaining the reasons behind the recall, you can take a look at the next part of this article on Similacrecall.com.
Why is it remembered Today?
We observed that Abbott has reacted by recalling the products since four children’s illnesses are related to their products that are affected through Salmonella as well as Cronobacter pathogens. Further, the research found that these products were made at Abbott’s Sturgis facility.
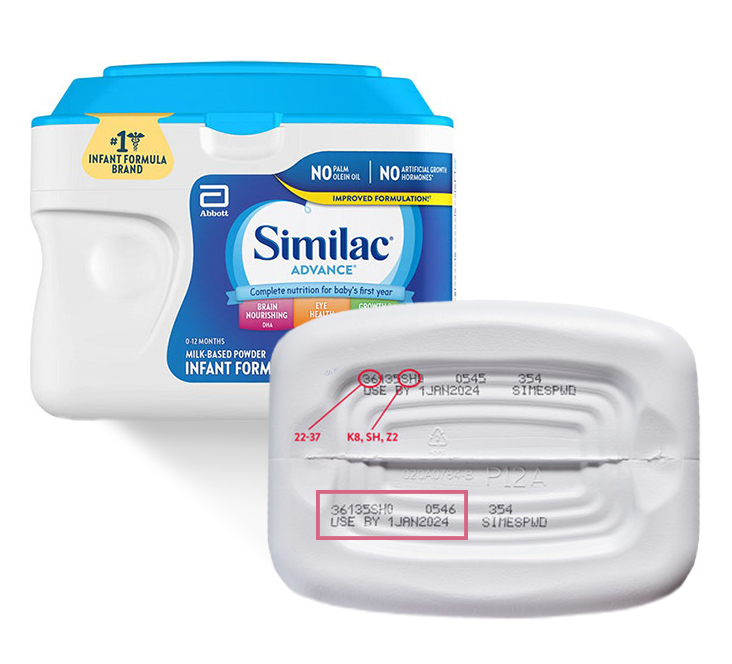
According to the most recent reports, the adverse effects of pathogens weren’t evident in Canadian infants, but have affected a handful of United States children. When analyzing the evidence for Similacrecall Com and its associated products, the FDA recommended that people avoid using products that contain codes 22-37 K8, SH , or Z2 and the date of suspension is April 1, 2022 or later.
What Should You Do If You Receive The Product That’s Affected?
The experts have suggested avoiding the use of certain products in case the product is under recall. You can also go to the website for either a refund or exchange. Additionally, you can seek assistance from medical experts or contact Similac customer service.
the Final Words
We formulated the basis for Similacrecall Com from the information available. Through a search on the subject we discovered it was because Abbott Laboratories had recalled their nutritional products, which included Similac after exposing it to harming newborn babies. Therefore, it is recommended to read this information as a guideline only and we are not verifying the authority of the person who wrote it. You can also conduct a search to get more details and find out more.